|
So take 3.5 – 3.0 and you get an intermediate answer of 0.5. First thing, the rules of algebra say to do the subtraction first, then the division. 
If you don’t learn this, you are going to lose points on your percent error calculations in lab reports… that’s a lot of points to lose! This is an extremely common case in a chemistry class for calculating things like percent change and percent error. You’ll also find the significant figures worksheet at the bottom of the page to get sig fig practice and master the topic.Ĭonsider a calculation like (3.5 – 3.0)/3.0 or in general (A-B)/B where A and B could be any number. 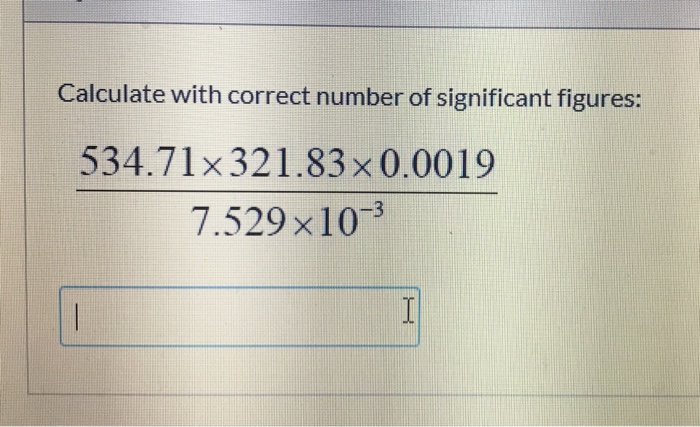
Everybody knows how to round, and sig figs tell us how many digits to round to. Again, we are just talking about rounding numbers. Sig figs is a fairly easy concept (really!) that usually looks difficult because it’s usually taught poorly. We have clear examples below that show you 3 simple rules to apply sig figs correctly in every situation. That is, should your final answer have 3 decimal places? Or none? How precise should your answer really be? Significant figures are used to express the idea of precision in science. Sig figs are used to determine where we should round. We all know how to round: 0-4 round down, and 5-9 rounds up. Rounding is a simple concept we usually learn in elementary school. In Plain English, significant figures are used for rounding numbers. The Significant Figures Worksheet covers sig figs, as they say, a topic found in chemistry classes that doesn’t exactly look like chemistry.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
